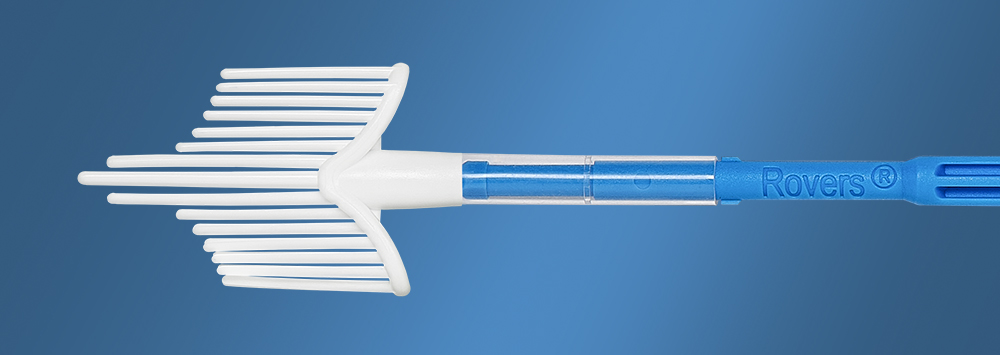
Cervical cancer is the fourth most common cancer and the fourth leading cause of cancer death among women world-wide. Screening for the detection of cervical cancer and pre-cancer has reduced the incidence and mortality rate of this cancer type. The Cervex-Brush® is the most widely used method for cervical cancer screening.
Optimal cervical cancer screening with a single device
A high quality examination is one of the important elements that determines efficacy of screening. The Cervex-Brush® was developed by Rovers® Medical Devices, in collaboration with women and physicians. Rovers’ specialists have experience in the development of medical devices for gynaecological examinations for over 20 years.
The Cervex-Brush® enables simultaneous collection of ectocervical, endocervical and transformation-zone cells with a single device. The brush is made up of well-defined semi-circular soft, flexible hairs for the optimal sample collection. The Cervex-Brush® can be used for HPV testing, conventional cytology and liquid-based cytology. The hydrophobic material of the brush facilitates the release of the cell material into the fluid or on to a glass slide.
Benefits
- Sweeps the portio and reaches well beyond the transformation-zone into the cervical canal for a rich cell yield.
- Significantly reduces the number of inadequate smears.1
- Avoids bleeding and pain which are normally associated with rigid spatulas and less considerate endocervical devices.
- The Rovers® Cervex-Brush® can be used for liquid-based as well as conventional cytology. The hydrophobic material of the brush facilitates the release of the cell material into the fluid.
- Optional detachable brush head can be supplied depending on the LBC method used.




